 Note a compound may be insoluble in one solvent yet fully miscible in another. 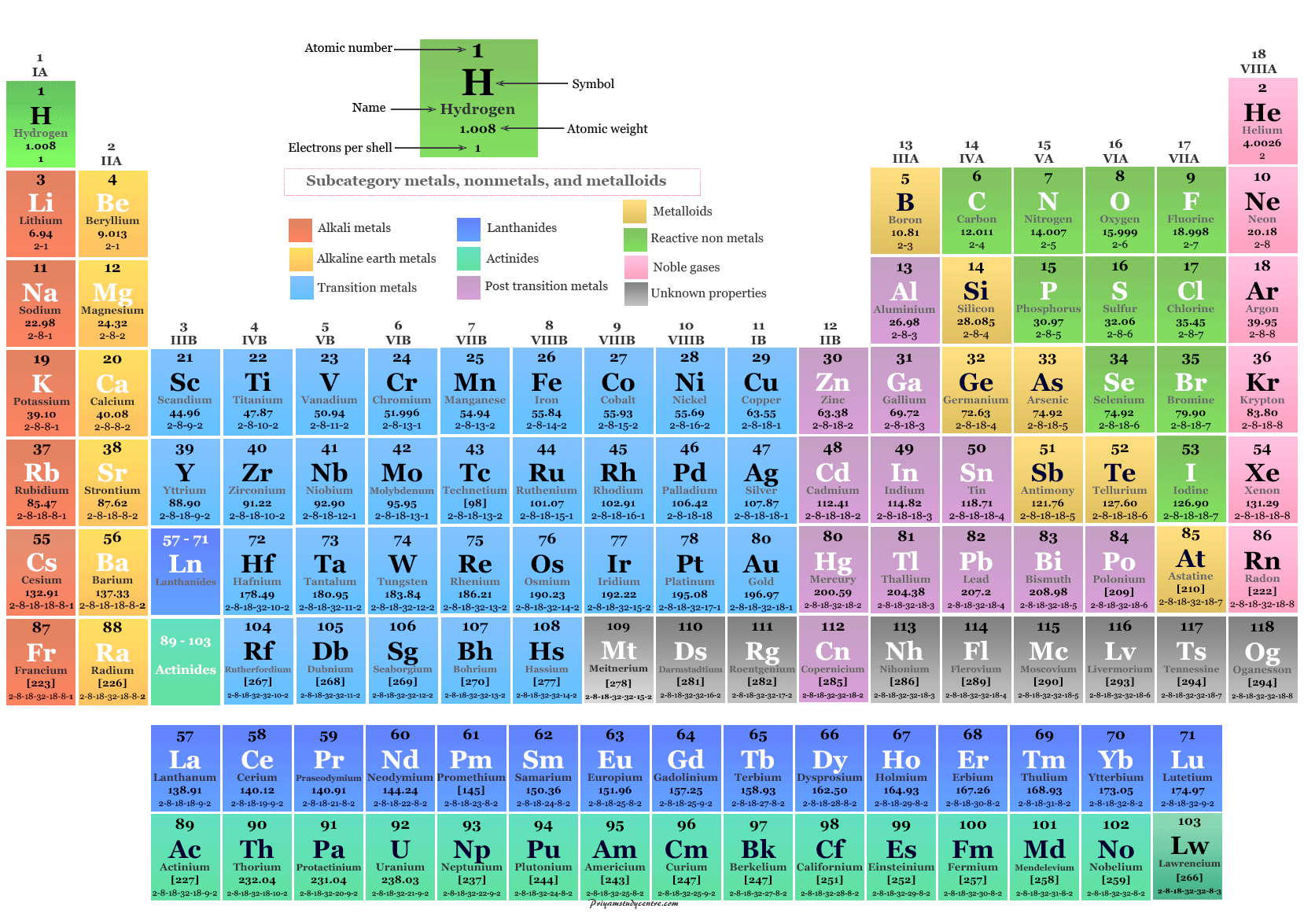
For example, very little silver chloride dissolves in water, so it is said to be insoluble in water. However, many substances are poorly soluble. 
It is rare for absolutely no solute to dissolve at all. Chapter 10: Chemical Bonding 10.1: Bonding Models and AIDS Drugs 10. Insoluble means incapable of dissolving in a solvent. In rechargeable batteries, such as many lithium-ion batteries, this chemical process is reversible and the internal structure different which allows the batteries to be recharged.ĭue to the ionic properties of salt water, scientists are now striving to exploit the ionic electricity-generating potential of salinity gradients where salt water and fresh water mix as a green source of energy generation for the future. 9.7: Electron Configurations and the Periodic Table 9.8: The Explanatory Power of the Quantum-Mechanical Model 9.9: Periodic Trends: Atomic Size, Ionization Energy, and Metallic Character Chapter 10. The zinc anode also acts as the battery’s container in zinc-carbon batteries so as it oxidizes during use, the contents can start to leak over time.įigure 4: A zinc-carbon dry cell battery (left) and alkaline battery (right). Moving left or right within a row, the atoms trend smaller. In single use, dry cell batteries, zinc is commonly used as the anode whilst manganese dioxide is a popular choice for the electrolyte cathode. Going up or down a column, elements are organized into groups which have similar chemical properties. As this ionic substance reacts with the electrodes it generates electrical current. 
In between the electrodes is an electrolyte liquid or gel that contains charged particles – ions. Heres the definition of the periodic table: The periodic table of elements is a table of chemical elements ordered based on their atomic number, electron. Batteries have two electrodes made of conductive material, the cathode which is the positive end where the electrical current leaves/electrons enter, and the anode where the electrical current enters/ electrons leave. Ionic properties are central to the function of batteries too. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
